
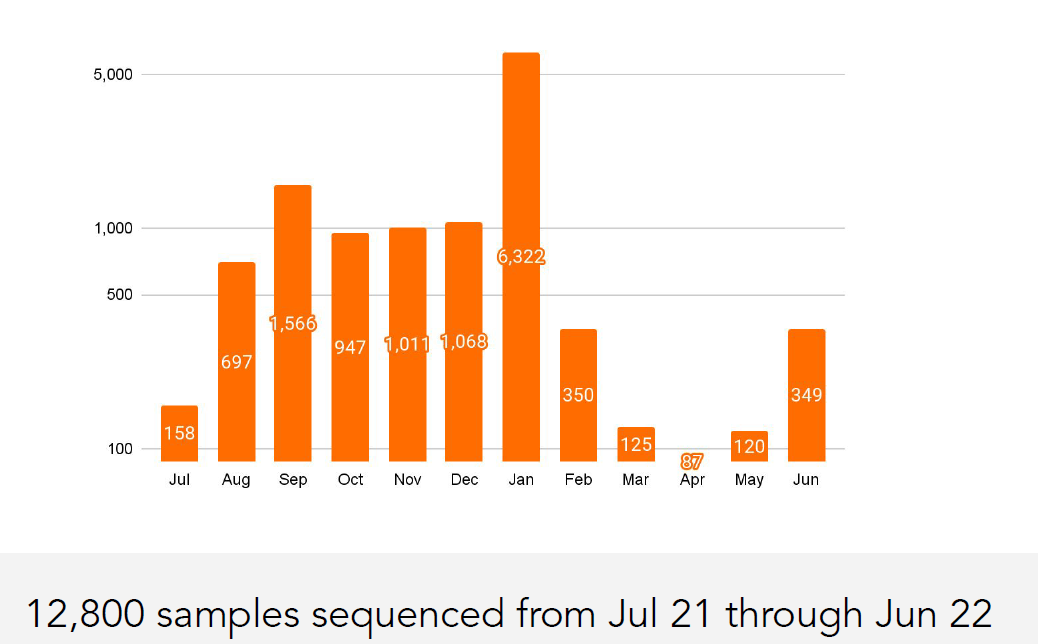
India is witnessing a fresh uptick in COVID-19 cases, with 257 active infections reported across nine states as of May 19, 2025. While the numbers remain relatively low compared to previous waves, health experts urge caution, particularly as new Omicron subvariants— JN.1 and its descendants LF.7 and NB.1.8 —drive infections
Where Are Cases Rising?
The states reporting the highest increases include:- Kerala: 95 cases (sharpest rise)
- Maharashtra: 56 cases
- Tamil Nadu: 66 cases
- Delhi, Gujarat, Haryana, Karnataka, Rajasthan, Sikkim: Smaller clusters.
Why Are Cases Rising Again?
Several factors contribute to the resurgence:- Waning immunity: Many people have not received booster doses.
- Relaxed preventive behavior: Mask-wearing and social distancing have declined.
- Seasonal effects: Increased travel and gatherings may be fueling transmission.
- Global trends: Countries like Singapore and Hong Kong have seen spikes due to Omicron subvariants.
What Symptoms Should You Watch For?
The dominant JN.1 subvariant typically causes mild symptoms, including:- Fever
- Sore throat
- Cough
- Fatigue
- Runny nose
- Occasional shortness of breath (in vulnerable individuals).
What Can You Do?
Health officials recommend:- Wearing masks in crowded places and enclosed spaces.
- Avoiding unnecessary travel if symptomatic.
- Getting tested promptly if symptoms appear.
- Following hand hygiene and respiratory etiquette.
- Staying up to date with vaccinations and booster doses.
The Bigger Picture: Lessons from the Pandemic
While India’s current COVID-19 situation is not alarming, it serves as a reminder that pandemic preparedness must remain a priority. The rise in cases highlights the importance of genomic surveillance, public health infrastructure, and community awareness in managing future outbreaks.As the world moves beyond the peak of the pandemic, vigilance—not panic—is key. Staying informed and adopting basic preventive measures can help curb the spread and protect vulnerable populations.





























 IndianWeb2.com is an independent digital media platform for business, entrepreneurship, science, technology, startups, gadgets and climate change news & reviews.
IndianWeb2.com is an independent digital media platform for business, entrepreneurship, science, technology, startups, gadgets and climate change news & reviews.